It's Time For Ethanol Honesty - with UPDATE
 |
Sparring with the new spate of anti-ethanol scatological slurs
 Marc Rauch |
Author of THE ETHANOL PAPERS
Exec. Vice President/Co-Publisher
THE AUTO CHANNEL
The April announcement by Joe Biden that E15 would be allowed to be sold year-round set off a frenzied spate of new attacks on the good fuel...the clean fuel...the 100% domestically produced fuel: ETHANOL.
TV and radio stations all over the country have been abuzz with contrived urgency concerning this decision as if the topic has never been discussed before. I can't say that I'm completely unhappy about this because these incidents have a silver lining: They open the door to present true information about ethanol and its benefits. The negative information works to bring the importance of alternative fuels back into the public eye for the opportunity to present honest, positive information.
For example, a news story from the "I-Team" at WGME, the CBS-affiliate in Portland, Maine, posed the question: "Is E15 ethanol fuel safe to use in every vehicle?"
 |
When the video of this story was posted on WGME's Facebook page it drew a swarm of local busy-bodies and know-it-alls to add their two cents. Proving that two cents is now worth far, far less than it was when the adage was first coined, the neighborly curbside advice was pure bovine scat. Having been alerted to the WGME Facebook post, I watched the video, read the comments, and then added some of my million-dollar advice (excuse my immodesty).
In response to my accurate counsel that all vehicles and internal combustion engines, regardless of size and age, could safely use E15 and higher blends, one yenta named Beth O'Connor replied to my ethanol advocacy by asking:
-
"Why reinvent the wheel (just stick with gasoline)?"
I chuckled to myself, and wrote back to her:
-
"Since alcohol fuels were the original fuels used in internal combustion engines, how is using cleaner, safer, healthier, less expensive ethanol reinventing the wheel?"
For those who may not be aware of it, Samuel Morey, the acknowledged inventor of the first internal combustion engine (1826) used an alcohol-based fuel. He did this nearly two hundred years ago - for those with weak vision, that's:
NEARLY 200 YEARS AGO.
A generation after Morey, a new kid on the block, Nicolaus Otto, used an alcohol-based fuel to power his versions of the internal combustion engine automobile starting in the 1860s. The petroleum oil formula we know of as gasoline (petrol in Britain terminology) didn't come into practical use for another couple of decades. Henry Ford's first automobile, the Quadricycle, used an alcohol-based fuel in 1896 instead of gasoline because his alcohol fuel was cheaper and easier to come by (there were no corner gasoline stations back then). Ford's Model T was designed to use neat ethanol as easily as gasoline. Tin Lizzie, as it was called, revolutionized the fledgling automobile industry, made owning a car an every man's dream come true, and it was the world's best-selling car for 20 years.
Incidentally, the EPA restrictions about E15 usage are absurd for a few reasons, such as the fact that when the EPA and government labs conducted their tests, they also tested E20 and found that E20 was just as safe to use as E15 and E10. In reality, all the hubbub shouldn't be about using E15 year-round, it should be about using E20.
In addition, Brazil has mandated the use of E15 and higher blends for ALL VEHICLES and spark-ignited internal combustion engines since 1978. They have mandated E20 and higher fuels since 1993, and for the past seven years, E27 has been mandated. They have the same cars, trucks, motorcycles, boats, and lawnmowers that we have in America.
The photo shown below comes from Honda's Brazilian Portuguese language website. It's a Honda gasoline powered generator - a small engine - that was sold and used in Brazil. It ran on E27 fuel. Notice on the engine sticker where else the generator was sold (in the U.S., where it meets EPA standards). If this generator can safely and efficiently run on E27 in Brazil, it can safely and efficiently run on E27 in America.
The cherry on the top of the cake is the fact that ethanol-gasoline blends were widely marketed throughout Great Britain for several decades beginning in the 1920s. The primary oil companies selling the E15 - E30 blends were industry giants Standard Oil (Esso) and Cities Service (Citgo). The argument that older and classic carbureted vehicles can't use E15 or higher blends are proven false because they could have, and probably did use higher ethanol-gasoline blends at different times without any issues. This is all documented in my reports:
The Yenta is not some ordinary Yenta
Beth O'Connor didn't reply to my historical notation about the early use of alcohol fuels, but she did respond. Ms. O'Connor wrote:
-
"... I have spent 10 years of my life researching this issue. Following the money was quite easy. I am quite pleased the work I did expanded the availability of E-0 in Maine. My efforts were fully
supported by a myriad of individuals from mechanics and automotive technicians to those who spend their lives striving for better air quality and environmental benefit."
|
|
This really caught my attention. After all, it's not every day that you come across someone who's proud to say they advocated the expanded availability of a pure poison like E0 gasoline.
 Beth O'Connor |
I call Ms. O'Connor's speech extravagantly comedic because her position is so wrong and her interpretation of the report so cockeyed that it's reminiscent of Groucho Marx' address to the fictional Freedonia Legislature in the 1933 movie "DUCK SOUP."
 |
I read through the conclusions, re-watched her video, scratched my head, went back to the report to try to find the specific 'horrific' details she refers to, then back again to her video, scratched my head some more, and went back to the report one more time.
Then I sent Beth O'Connor the following:
"Well, Beth, 10 years research is pretty good, congratulations. But, it's not the more than three decades of my related research, investigation, and hands-on experimentation. Perhaps in 20 years, when you graduate to the major leagues, the truth about ethanol's benefits will become apparent to you.
"I watched the video of you from 2019. In it, you say that an "extensive and overdue report from the National Department of Environmental Protection" states that in no uncertain terms that including ethanol into the U.S. gas supply is wreaking havoc on our environment. What is the National Department of Environmental Protection? We don't have a "National Department of Environmental Protection." We have the Environmental Protection Agency, and many states have separate environmental agencies, but there's no "National Department of Environmental Protection." Did you simply misspeak, or is this an example of how you get confused over words and meanings. Maybe this is why you think that some governmental body has issued such a dire warning against ethanol?
"In fact, I read the report you referred to, the Second Triennial Report to Congress. Where in the report do you think that the EPA states "in no uncertain terms that including ethanol in the U.S. gasoline supply is wreaking havoc on our environment?" I ask because it doesn’t say that, nor does it imply it. You over-exaggerated the conclusions of the report…you lied about it.
The EPA's comments about the effects of growing corn would probably be the same comments that they would make (if asked) about the growing of any crop on previously un-used land. Are you suggesting that the growing of crops to feed our growing population should stop? Should we stop cultivating any new land? Should we not re-use land that has already been used?
"Moreover, you make the puerile statement that "The powerful corn lobby in Washington greases palms and purchases politicians to sweep the horrors of this product under the rug." You have the unmitigated gall to say this when the truth is that the petroleum oil lobby has spent hundreds of millions of dollars - maybe billions of dollars - since the 1920s to buy politicians and hide the real horrors of tetraethyl lead, benzene, and proven dangerous emissions from gasoline. You single out Archer-Daniels-Midland and Monsanto as evil conglomerates. ADM and Monsanto are mom-and-pop companies by comparison to the oil companies and their affiliated chemical companies like DuPont. You should be embarrassed for making these remarks.
"You write that your "efforts were fully supported by a myriad of individuals from mechanics and automotive technicians to those who spend their lives striving for better air quality and environmental benefit." Yeah, well my position is fully supported by real mechanics, auto technicians, and some of the world's greatest scientists and inventors that actually know what they're talking about.
"You have the impudence to say that you are “quite pleased the work I did expanded the availability of E-0 in Maine.” E0 … ethanol-free gasoline … is poison, 100% poison. You are murdering the people of Maine. That’s what you are proud of?
"Beth, I’m going to tell you straight up that E10 isn’t perfect, E15 isn’t perfect, even E85 is not perfect They have big, big problems. You know what those problems are? I’ll tell you if you promise NOT to keep it a secret: E10 has about 90% gasoline in it. E15 has about 85% gasoline in it. And even E85 has anywhere from about 17% to 49% gasoline in it! You can clean open skin cuts with ethanol. You can drink and gargle with ethanol. You can clean sensitive electronic equipment with ethanol. You can preserve food with ethanol. If you tried this with gasoline you'd be dead and have gummed-up electronic equipment.
"On the two occasions that I was invited to Washington, DC to provide testimony to Congressional Committees, I presented real information, not hysterical, out-of-context mischaracterizations like you did. I also conducted actual simple experiments onsite to support my words.
"Regarding your mention in the video of the Gulf of Mexico hypoxia problem, as I replied to you in a previous exchange, hypoxia is a condition that exists in many parts of the world, and it typically has nothing to do with the growth of ethanol-related crops or the production of ethanol fuel. The hypoxia problem in the Gulf of Mexico was initially identified in the 1950s, a half century before the passing of the Renewable Fuel Standard and the regular production and use of ethanol fuels. If you or anyone else singles out the growing of any ethanol crop as the reason for the problem, you are making a fool out of yourself. It would be far better to blame golf for the growth of hypoxia than the growing of corn. There are thousands of golf courses in the states that have streams and rivers that run into the Gulf of Mexico. All of these golf courses use fertilizers and other chemicals year-round to keep their acres and acres of grass green. These chemicals all run off into the rivers and streams. On top of this are the vast residential and commercial developments, as well as academic campuses. And corn isn’t the only crop grown in these states.
"You ignorantly blame ethanol for lung disease and asthma. Ethanol is not what has caused lung disease and asthma; these diseases are largely caused by gasoline and petroleum diesel fuel emissions. These fuel emissions have also contributed to the rise in mental defects. All gasoline contains benzene. Even the oil industry has stated that there is no acceptable amount of benzene in the air, and yet the only way the oil industry can increase the octane level of gasoline without an alcohol component would be to increase the amount of benzene or return to the use of tetraethyl lead. Is that what you want, to increase benzene and TEL? Is that what your 10 years of research has taught you?"
• UPDATE: On May, 1, 2022 Beth O'Connor responded to me. CLICK HERE.
 Click to enlarge |
(This passage can be found in chapter 5, TRIGGERS, sub-section POLLUTION. The book was written in 1995, prior to any measurable ethanol-gasoline fuel use in the United States. It's also worth noting that the passage makes no mention whatsoever of ethanol - or any alcohol - being a contributory factor in creating NO2.)
MEANWHILE, IN TEXAS
Houston's FOX-affiliate KRIV aired a story similar to the Maine TV station story. In this case, the question was "Will lifting the E15 gasoline ban really save you money"? It was posed by their consumer reporter Heather Sullivan on a two-part segment called Sullivan's Smart Sense.
Part 1
Part 2
Ms. Sullivan rattles off a few factoids about ethanol fuels and then says that the ban on E15 is because in warmer weather the extra ethanol can create smog. She introduces a professor from the University of Houston to explain this issue and more. The smog issue is idiotic and I'll get to that issue shortly. But first, I want to expose what I think is a very underhanded part of the anti-ethanol argument, although Ms. Sullivan may not be doing it intentionally.
Here it is: Ms. Sullivan and her professor talk about the price differences and miles-per-gallon differences between E10 and E15. They both treat the issues as if they are inconsequential and almost interchangeable. Ms. Sullivan reports that the EPA says that using E10 loses about 3% to 4% mpg, and that E15 loses about 4% to 5% mpg. She shows on the pump that the price of E15 is five cents per gallon cheaper (she mistakenly says 3 cents) than E10, and so she says it's a wash. The prices are $3.58 for E15 and $3.63 for E10 (I rounded off the prices for simplicity). During the professor's comments, he pretty much says the same thing.
However, neither Sully nor the professor actually say what the price differences are compared to. Why this omission is shady and underhanded is that the price of E0 can be significantly higher than E10 and E15. The filling station that they are at in the video is a Murphy USA station. This Murphy station does not sell E0. I went online and looked for stations in the Houston area that do sell E0. I found one called Fuel Wise. The price right now for regular 87 octane E0 is $4.89 per gallon. Therefore, the price difference between Fuel Wise's E0 and Murphy's E10 is $1.26 per gallon. This means that the E10 is a 26% savings versus the cost of E0. If E10 loses 4% mpg compared to E0 then there is still a net savings of a little more than 20% per gallon. Since the E15 is a nickel cheaper than E10 it means the cost difference between E15 and E0 is $1.31, a 27% savings.
There's more to the story: E15 is not an 87 octane fuel, it's 88 octane. So a comparison of the price of E15 to the price of regular 87 octane E0 is not exactly even. We have to look at the cost of a higher octane E0, for example a mid-range 89 octane E0.
Unfortunately, the Fuel Wise station I called doesn't sell a mid-octane E0, but from previous experience, I estimate that the price of 89 octane E0 could be about 40 cents more than the 87 octane. If we split the amount, we can add 20 cents to the price of the E0 to make the comparison with E15 fairer. This increases the saving between E15 and a comparable E0 to $1.51 per gallon, a 30% savings. Based on this price vs. mpg comparison, if you lose 5% mpg by using E15, you still have a net gain of 25%. Since the physical effort of pumping E15 into your fuel tank is no greater than the effort expended to pump in E10 or E0, it means that lifting the ban on E15 does save you even more money than E10 already saves you. The question posed by Heather Sullivan has now been answered: E15 will save consumers even more money than E10!
Oh, but wait, we still have the professor and the issue of smog to deal with.
 Ramanan Krishnamoorti |
If someone asks if Heather Sullivan could find a more highly credentialed person to interview you'd have to say "forgeddaboutit" in a Brooklyn accent.
The problem is the smog. Dr. K says that the volatility of ethanol causes smog; that E15's evaporative volatility will create more pollution.
Here's the thing...smog is not new. The blindingly dense infamous London fog was caused by smog from all the coal and wood fires used to heat, light, cook, and power the manufacturing plants of the industrial revolution. Smoke from the fires, plus the natural fog was linguistically combined to give us the word "smog." This type of smog is referred to as "Industrial Smog," and it began to be serious in the 1700s, almost 300 years ago. Industrial Smog was not, and is not caused by ethanol burning.
In the early 1940s, a second type of smog, "Photochemical Smog," was given the nickname "LA Smog" because it had become so bad in Los Angeles, California. Photochemical Smog is primarily caused by emissions from gasoline and diesel fuel-powered internal combustion engines. The emissions create what is called low-level ozone. Photochemical Smog can occur anywhere where there is the right combination of low-level ozone and lots of sunshine. With so much attention being given to the "Tinseltown" glamour of Hollywood it was more fun to name it LA Smog rather than New York Smog or Chicago Smog.
For the conditions of LA Smog to become common enough to be identified with the nickname LA SMOG, it means that the situation had to have already been present and noticed for an extended period of time. Cars and trucks didn't just suddenly appear in Los Angeles in the 1940s. According to Smithsonian Magazine, "L.A.’s population of about 600,000 at the start of the 1920s more than doubled during the decade. The city’s cars would see an even greater increase, from 161,846 cars registered in L.A. County in 1920 to 806,264 registered in 1930. In 1920 Los Angeles had about 170 gas stations. By 1930 there were over 1,500." From other sources, we know that by 1940 the number of automobiles in L.A. County was well over one million.
The significance to all this is that ethanol fuel was not being used in internal combustion engines in America during the 1920s, nor 1930s, nor the 1940s, '50s, '60s, '70s, '80s, or '90s.
A 5.7% ethanol-gasoline blend (E5) began being used in California after 2003 when MTBE was banned from gasoline (poisonous MTBE was the oil industry's brilliant go-to suggestion to eliminate engine knock after tetraethyl lead was finally banned).
Therefore, from the beginning of the Roaring 20s and the introduction of leaded gasoline needed to eliminate engine knock in the new high compression automobile engines of the time, to an E5 blend after 2003, more than 80 years passed before ethanol was regularly added to gasoline. Yet in all those years of non-ethanol fuel availability, the conditions of Photochemical Smog in Southern California became so terrible that it engendered a universally known nickname and it became the butt of endless late-night TV talk show jokes.
The Renewable Fuel Standard wasn't enacted until 2005, and E10 wasn't put into regular use until about 2010. While society can, and has, laid blame for smog on gasoline and petroleum diesel fuel, ethanol was not the cause for smog - not Industrial Smog and not Photochemical Smog. But there are people like Ramanan Krishnamoorti who claim something different.
Dr. K says that the volatile evaporative nature of ethanol causes more smog. And he does have unbelievable professional and academic credentials!!!
 |
The contributing chemical components of low-level ozone and Photochemical Smog are nitrogen oxides, unburned hydrocarbons, and volatile organic compounds (VOCs). Nitrogen oxides are emitted from the combustion of gasoline and petroleum diesel fuel. Unburned hydrocarbons are a result of the inefficient burning of petroleum oil fuels (gasoline and diesel). Gasoline's burning inefficiency is why dark smoke is emitted. Ethanol burns very efficiently which is why it produces no dark smoke. What does the comparative burning of gasoline and ethanol look like? Watch the video just below to see.
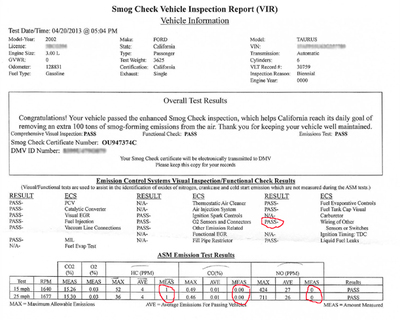
Does the combustion of ethanol produce any nitric oxide emissions? Some reports say yes. My practical, hands-on experience is that it doesn't. My experience is based on the smog test that my non-flex fuel 2002 Ford Taurus went through. I've written about this several times. I purchased the vehicle used in 2011 for the specific purpose of testing various ethanol-gasoline blends above E10. I ran the car on every level possible from 10% (E10) to as close to 85% (E85) as I could. In 2012, I had to submit my car for a mandatory smog test. The day I drove the car into the shop, I was running on about E40. The document below shows the results.
As you can read, the state's smog test is to determine the amount of particulate matter emitted by automobile engines. It measures three categories: Hydrocarbons (HC), Carbon Monoxide (CO), and Nitrogen Oxide (NO).
In order to pass the smog test, my car had to at least meet allowable "parts per million" (PPM) levels at two different engine speeds, as set for this specific type of vehicle:
HC - 52/36 The average passable score was 4/4 My car's score 1/1
CO - 49/46 The average passable score was 0.01/0.01 My car's score 0.00/0.00
NO - 424/711 The average passable score was 27/26 My car's score 0/0
No nitrogen oxide emissions were detected!
The other primary chemical component, volatile chemical compounds, contain benzene, toluene, and xylene. These substances are collectively called "aromatics," and are considered dominant contributors to the creation of low-level ozone. Ethanol does not contain benzene, toluene, or xylene. Gasoline cannot be produced without including benzene, toluene, and xylene.
To create an anti-knock ethanol-free gasoline that does not contain tetraethyl lead or MTBE more aromatics must be used. These are poisons. How poisonous? In a 1948 TOXICOLOGICAL REVIEW conducted by the American Petroleum Institute (the oil industry's main lobbying organization), their study "summarizes the best available information on the properties, characteristics, and toxicology of benzene." The most decisive part of the report is the part in which they give what they believe is a safe ppm (parts per million) amount of benzene in the air we breathe. The report states:
"Inasmuch as the body develops no tolerance to benzene, and as there is a wide variation in individual susceptibility, it is generally considered that the only absolutely safe concentration for benzene is zero."
The complete API report can be found by CLICKING HERE.
An interesting way to imagine the difference between gasoline and ethanol emissions consider what would happen if you put yourself in a sealed, unventilated one-car garage with an internal combustion engine vehicle running on just E0 for a couple of hours. You'd be dead. On the other hand, pure ethanol burns so cleanly that the people can be in a closed room burning ethanol (without any extraneous type of equipment to clean the emissions) and survive the experience quite handily. Ethanol is used safely for indoor lighting, heating, and cooking.
So, if ethanol doesn't contain any of the aromatics, it burns clean without emitting carbon debris, and higher levels of ethanol-gasoline blends don't emit nitrogen oxides, does this mean that the Michael Jordan of the academic chemistry world is wrong?
I definitely think it does!
To screw a tight lid on this discussion, I go to the final part of Dr. K's anti-ethanol postulation. He says that ethanol is so evaporative that just the few extra seconds involved in topping off your fuel tank with E15 could release more fumes into the air, thereby creating more smog, thereby making ethanol-gasoline blends more polluting than non-ethanol gasoline.
The problem with Ramanan Krishnamoorti's hypothesis is that gasoline vaporizes at a lower temperature than ethanol; GASOLINE EVAPORATES FASTER THAN ETHANOL. If evaporation is the catalyst to causing smog, then ethanol is a hero, not a villain. And if a few seconds of a gas cap being open is so precious in the fight to lower smog creation, then we should demand that E15 replaces all E10 because there is 50% more of the less volatile ethanol. What's more, E20, E30, E40, E50, all the way up to E85 should be far more preferable than E10, E15, and E0. Better still, remove all gasoline completely and utilize a different method to overcome cold-starting problems; use methods similar to those used in diesel fuel-powered engines.
THE CLOSING ACT
I hate to kick a professor when he's down, but Dr. Krishnamoorti made a couple more mistakes. He talks about water forming in a fuel tank, and he blames this on ethanol. Water will form in a fuel tank because of condensation - good old natural condensation. It happens whether or not any ethanol is present, and the presence of ethanol does not heighten the possibility of condensation occurring. Conversely, ethanol aids in removing any water in a fuel tank, so it is beneficial, not detrimental.
He also says that in a real free market that gasoline would be less expensive than ethanol because of ethanol subsidies, but there are no ethanol subsidies. There are, however, plenty of gasoline subsidies. Moreover, there is no free market when it comes to petroleum oil and its derivative fuels. This has never been a free market. The oil market is tenaciously controlled and manipulated by greedy foreign and domestic people.
I truly respect people who commit themselves to years of academic study, but I don't respect those who are willing to throw their integrity away and sell themselves to advance someone else's lies. I have no problem with people using their name, fame, and credibility to endorse a product or issue for money, as long as they are truthful and the issue/product is righteous. I have absolutely no idea if Ramanan Krishnamoorti has taken any money from the oil industry to promote ethanol misinformation. And I'm not implying that he has, but I'd be willing to bet that the University, or the departments that Krishnamoorti has worked for have received funding from the oil industry. The funding, if any, may not have come with a proviso that false ethanol information be spread, it may just be an instance where some people on the receiving end don't want to risk losing future funding, so they back off from being forthright and controversial about the facts.
By the way, I have conveyed my feeling to Dr. Krishnamoorti in a private email. I told him he should be ashamed of his statements in the interview.
For more information about the issues I've addressed regarding Ramanan Krishnamoorti's claims, please read the following:
• Just Because It's in Popular Science Magazine That Doesn't Mean It's True
• Ethanol Does NOT Suck Water Out Of The Air
• It's Time To Rethink The Value Of PhD
• Open Letter to Kate McAlpine and Michigan Engineer News Center on Ethanol Fuel
ABOUT THE AUTHOR
Marc J. Rauch is a multi-award winning TV/film writer, producer and director and has been a broadcasting and marketing executive for more than 50 years. In 1995, Marc wrote and published "MARCETING - The Book On Effective Advertising and Promotion." This book was updated and re-published in 2005 with additional chapters covering the Internet and online marketing.
In 1987, Marc co-founded The Auto Channel television network, and TheAutoChannel.com a few years later. Marc serves as the company's Executive Vice President and Co-Publisher. TheAutoChannel.com is the Internet's largest and oldest automotive information resource, with more than 1.5 million pages of content. Marc is a leading advocate of alternative fuels and energy; writing, lecturing and making numerous broadcast appearances regarding these issues. He's traveled to Taiwan to speak at the Asia-Pacific Economic Cooperation Forum on behalf of the U.S. Department of Agriculture, he's presented testimony on the use of ethanol before Congressional debriefings in Washington on two occasions, was a keynote speaker at the 2016 Australian National Biofuels Conference, and was a featured speaker at the 2018 National Ethanol Conference in San Antonio, Texas. In 2019, Marc wrote and published "THE ETHANOL PAPERS," a 600-page book that presents the whole story on ethanol fuels. In 2021, Marc published his extended essay "YES, TIN LIZZIE WAS AN ALCOHOLIC" about Henry Ford's Model T in book form and is available from AMAZON BOOKS.