H2 And Fuel-Cell Tutorial
SEE ALSO: Hydrogen Fuel Cell Vehicle Archive - News, Articles, Opinion, Tutorials and Video 1996-Today
Originally Published 2/24/2010
AUTO CENTRAL - February 24, 2010: A potential future replacement for a gasoline engine in extended range Electric Vehicles will be the fuel cell which converts a non-petroleum fuel into electricity to recharge batteries. Fuel cells are classified primarily by the kind of electrolyte they employ. This classification determines the kind of chemical reactions that take place in the cell, the kind of catalysts required, the temperature range in which the cell operates, the fuel required, and other factors. These characteristics, in turn, affect the applications for which these cells are most suitable. There are several types of fuel cells currently under development, each with its own advantages, limitations, and potential applications.
How Fuel Cells Work
Fuel cells work like batteries, but they do not run down or need recharging. They produce electricity and heat as long as fuel is supplied. A fuel cell consists of two electrodes—a negative electrode (or anode) and a positive electrode (or cathode)—sandwiched around an electrolyte. A fuel, such as hydrogen, is fed to the anode, and air is fed to the cathode. In a polymer electrolyte membrane fuel cell, a catalyst separates hydrogen atoms into protons and electrons, which take different paths to the cathode. The electrons go through an external circuit, creating a flow of electricity. The protons migrate through the electrolyte to the cathode, where they reunite with oxygen and the electrons to produce water and heat
class="entity entity-paragraphs-item paragraphs-item-paragraphs-sp-heading psp section-subheading">Types of Fuel CellsAlthough the basic operations of all fuel cells are the same, special varieties have been developed to take advantage of different electrolytes and serve different application needs. The fuel and the charged species migrating through the electrolyte may be different, but the principle is the same. An oxidation occurs at the anode, while a reduction occurs at the cathode. The two reactions are connected by a charged species that migrates through the electrolyte and electrons that flow through the external circuit.
Polymer Electrolyte Membrane Fuel Cells
Polymer electrolyte membrane (PEM) fuel cells, also called proton exchange membrane fuel cells, use a proton-conducting polymer membrane as the electrolyte. Hydrogen is typically used as the fuel. These cells operate at relatively low temperatures and can quickly vary their output to meet shifting power demands. PEM fuel cells are the best candidates for powering automobiles. They can also be used for stationary power production. However, due to their low operating temperature, they cannot directly use hydrocarbon fuels, such as natural gas, liquefied natural gas, or ethanol. These fuels must be converted to hydrogen in a fuel reformer to be able to be used by a PEM fuel cell.
Direct-Methanol Fuel Cells
The direct-methanol fuel cell (DMFC) is similar to the PEM cell in that it uses a proton conducting polymer membrane as an electrolyte. However, DMFCs use methanol directly on the anode, which eliminates the need for a fuel reformer. DMFCs are of interest for powering portable electronic devices, such as laptop computers and battery rechargers. Methanol provides a higher energy density than hydrogen, which makes it an attractive fuel for portable devices.
Alkaline Fuel Cells
Alkaline fuel cells use an alkaline electrolyte such as potassium hydroxide or an alkaline membrane that conducts hydroxide ions rather than protons. Originally used by the National Aeronautics and Space Administration (NASA) on space missions, alkaline fuel cells are now finding new applications, such as in portable power.
Phosphoric Acid Fuel Cells
Phosphoric acid fuel cells use a phosphoric acid electrolyte that conducts protons held inside a porous matrix, and operate at about 200°C. They are typically used in modules of 400 kW or greater and are being used for stationary power production in hotels, hospitals, grocery stores, and office buildings, where waste heat can also be used. Phosphoric acid can also be immobilized in polymer membranes, and fuel cells using these membranes are of interest for a variety of stationary power applications.
Molten Carbonate Fuel Cells
Molten carbonate fuel cells use a molten carbonate salt immobilized in a porous matrix that conducts carbonate ions as their electrolyte. They are already being used in a variety of medium-to-large-scale stationary applications, where their high efficiency produces net energy savings. Their high-temperature operation (approximately 600°C) enables them to internally reform fuels such as natural gas and biogas.
Solid Oxide Fuel Cells
Solid oxide fuel cells use a thin layer of ceramic as a solid electrolyte that conducts oxide ions. They are being developed for use in a variety of stationary power applications, as well as in auxiliary power devices for heavy-duty trucks. Operating at 700°C–1,000°C with zirconia-based electrolytes, and as low as 500°C with ceria-based electrolytes, these fuel cells can internally reform natural gas and biogas, and can be combined with a gas turbine to produce electrical efficiencies as high as 75%.
In addition to electricity, fuel cells produce heat. This heat can be used to fulfill heating needs, including hot water and space heating. Combined heat and power fuel cells are of interest for powering houses and buildings, where total efficiency as high as 90% is achievable. This high-efficiency operation saves money, saves energy, and reduces greenhouse gas emissions.
This special class of fuel cells produces electricity from hydrogen and oxygen, but can be reversed and powered with electricity to produce hydrogen and oxygen. This emerging technology could provide storage of excess energy produced by intermittent renewable energy sources, such as wind and solar power stations, releasing this energy during times of low power production.
How All Types of Fuel Cells Work From The U.S. Department of Energy
Polymer Electrolyte Membrane (PEM) Fuel Cells
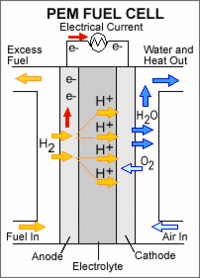 |
A PEM fuel cell consists of a polymer electrolyte membrane sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air is channeled to the cathode on the other side of the cell. 2. At the anode, a platinum catalyst causes the hydrogen to split into positive hydrogen ions (protons) and negatively charged electrons. 3. The Polymer Electrolyte Membrane (PEM) allows only the positively charged ions to pass through it to the cathode. The negatively charged electrons must travel along an external circuit to the cathode, creating an electrical current. 4. At the cathode, the electrons and positively charged hydrogen ions combine with oxygen to form water, which flows out of the cell."
Polymer electrolyte membrane (PEM) fuel cells—also called proton exchange membrane fuel cells—deliver high-power density and offer the advantages of low weight and volume, compared with other fuel cells. PEM fuel cells use a solid polymer as an electrolyte and porous carbon electrodes containing a platinum catalyst. They need only hydrogen, oxygen from the air, and water to operate and do not require corrosive fluids like some fuel cells. They are typically fueled with pure hydrogen supplied from storage tanks or on-board reformers.
Polymer electrolyte membrane fuel cells operate at relatively low temperatures, around 80°C (176°F). Low-temperature operation allows them to start quickly (less warm-up time) and results in less wear on system components, resulting in better durability. However, it requires that a noble-metal catalyst (typically platinum) be used to separate the hydrogen's electrons and protons, adding to system cost. The platinum catalyst is also extremely sensitive to CO poisoning, making it necessary to employ an additional reactor to reduce CO in the fuel gas if the hydrogen is derived from an alcohol or hydrocarbon fuel. This also adds cost. Developers are currently exploring platinum/ruthenium catalysts that are more resistant to CO.
PEM fuel cells are used primarily for transportation applications and some stationary applications. Due to their fast startup time, low sensitivity to orientation, and favorable power-to-weight ratio, PEM fuel cells are particularly suitable for use in passenger vehicles, such as cars and buses.
A significant barrier to using these fuel cells in vehicles is hydrogen storage. Most fuel cell vehicles (FCVs) powered by pure hydrogen must store the hydrogen on-board as a compressed gas in pressurized tanks. Due to the low-energy density of hydrogen, it is difficult to store enough hydrogen on-board to allow vehicles to travel the same distance as gasoline-powered vehicles before refueling, typically 300–400 miles. Higher-density liquid fuels, such as methanol, ethanol, natural gas, liquefied petroleum gas, and gasoline, can be used for fuel, but the vehicles must have an on-board fuel processor to reform the methanol to hydrogen. This requirement increases costs and maintenance. The reformer also releases carbon dioxide (a greenhouse gas), though less than that emitted from current gasoline-powered engines.
Direct Methanol Fuel Cells
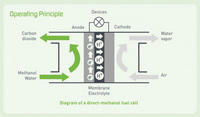 |
Direct methanol fuel cells do not have many of the fuel storage problems typical of some fuel cells because methanol has a higher energy density than hydrogen—though less than gasoline or diesel fuel. Methanol is also easier to transport and supply to the public using our current infrastructure because it is a liquid, like gasoline.
Direct methanol fuel cell technology is relatively new compared with that of fuel cells powered by pure hydrogen, and DMFC research and development is roughly 3–4 years behind that for other fuel cell types.
Alkaline Fuel Cells
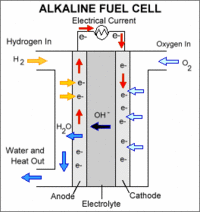 |
An AFC consists of an alkaline electrolyte, typically potassium hydroxide (KOH), sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air is channeled to the cathode on the other side of the cell. 2. At the anode, a platinum catalyst causes the hydrogen to split into positive hydrogen ions (protons) and negatively charged electrons. 3. The positively charged hydrogen ions react with hydroxyl (OH-) ions in the electrolyte to form water. 4. The negatively charged electrons cannot flow through the electrolyte to reach the positively charged cathode, so they must flow through an external circuit, forming an electrical current. 5. At the cathode, the electrons combine with oxygen and water to form the hydroxyl ions that move across the electrolyte toward the anode to continue the process."
Alkaline fuel cells (AFCs) were one of the first fuel cell technologies developed, and they were the first type widely used in the U.S. space program to produce electrical energy and water on-board spacecrafts. These fuel cells use a solution of potassium hydroxide in water as the electrolyte and can use a variety of non-precious metals as a catalyst at the anode and cathode. High-temperature AFCs operate at temperatures between 100°C and 250°C (212°F and 482°F). However, newer AFC designs operate at lower temperatures of roughly 23°C to 70°C (74°F to 158°F)
AFCs' high performance is due to the rate at which chemical reactions take place in the cell. They have also demonstrated efficiencies near 60% in space applications.
The disadvantage of this fuel cell type is that it is easily poisoned by carbon dioxide (CO2). In fact, even the small amount of CO2 in the air can affect this cell's operation, making it necessary to purify both the hydrogen and oxygen used in the cell. This purification process is costly. Susceptibility to poisoning also affects the cell's lifetime (the amount of time before it must be replaced), further adding to cost.
Cost is less of a factor for remote locations, such as space or under the sea. However, to effectively compete in most mainstream commercial markets, these fuel cells will have to become more cost-effective. AFC stacks have been shown to maintain sufficiently stable operation for more than 8,000 operating hours. To be economically viable in large-scale utility applications, these fuel cells need to reach operating times exceeding 40,000 hours, something that has not yet been achieved due to material durability issues. This obstacle is possibly the most significant in commercializing this fuel cell technology.
Phosphoric Acid Fuel Cells
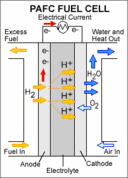 |
A PAFC consists of liquid phosphoric acid electrolyte sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air is channeled to the cathode on the other side of the cell. 2. At the anode, a platinum catalyst causes the hydrogen to split into positive hydrogen ions (protons) and negatively charged electrons. 3. The phosphoric acid electrolyte allows only the positively charged ions to pass through it to the cathode. The negatively charged electrons must travel along an external circuit to the cathode, creating an electrical current. 4. At the cathode, the electrons and positively charged hydrogen ions combine with oxygen to form water, which flows out of the cell."
Phosphoric acid fuel cells use liquid phosphoric acid as an electrolyte—the acid is contained in a Teflon-bonded silicon carbide matrix—and porous carbon electrodes containing a platinum catalyst. The chemical reactions that take place in the cell are shown in the diagram to the right.
The phosphoric acid fuel cell (PAFC) is considered the "first generation" of modern fuel cells. It is one of the most mature cell types and the first to be used commercially. This type of fuel cell is typically used for stationary power generation, but some PAFCs have been used to power large vehicles such as city buses.
PAFCs are more tolerant of impurities in fossil fuels that have been reformed into hydrogen than PEM cells, which are easily "poisoned" by carbon monoxide because carbon monoxide binds to the platinum catalyst at the anode, decreasing the fuel cell's efficiency. They are 85% efficient when used for the co-generation of electricity and heat but less efficient at generating electricity alone (37%–42%). This is only slightly more efficient than combustion-based power plants, which typically operate at 33%–35% efficiency. PAFCs are also less powerful than other fuel cells, given the same weight and volume. As a result, these fuel cells are typically large and heavy. PAFCs are also expensive. Like PEM fuel cells, PAFCs require an expensive platinum catalyst, which raises the cost of the fuel cell.
Molten Carbonate Fuel Cells
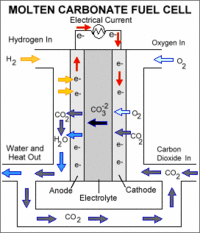 |
A MCFC consists of an electrolyte, typically a molten carbonate salt mixture suspended in a ceramic matrix, sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air, carbon dioxide, and electricity (electrons from the fuel cell circuit) are channeled to the cathode on the other side of the cell. 2. At the cathode, the oxygen, carbon dioxide, and electrons react to form positively charged oxygen ions and negatively charged carbonate ions. 3. The carbonate ions move through the electrolyte to the anode. 4. At the anode, a catalyst causes the hydrogen combine with the carbonate ions, forming water and carbon dioxide and releasing electrons. 5. The electrolyte does not allow the electrons to pass through it to the cathode, forcing them to flow through an external circuit to the cathode. This flow of electrons forms an electrical current. 6. The carbon dioxide formed at the anode is often recycled back to the cathode."
Molten carbonate fuel cells (MCFCs) are currently being developed for natural gas and coal-based power plants for electrical utility, industrial, and military applications. MCFCs are high-temperature fuel cells that use an electrolyte composed of a molten carbonate salt mixture suspended in a porous, chemically inert ceramic lithium aluminum oxide (LiAlO2) matrix. Because they operate at extremely high temperatures of 650°C (roughly 1,200°F) and above, non-precious metals can be used as catalysts at the anode and cathode, reducing costs.
Improved efficiency is another reason MCFCs offer significant cost reductions over phosphoric acid fuel cells (PAFCs). Molten carbonate fuel cells can reach efficiencies approaching 60%, considerably higher than the 37%–42% efficiencies of a phosphoric acid fuel cell plant. When the waste heat is captured and used, overall fuel efficiencies can be as high as 85%.
Unlike alkaline, phosphoric acid, and polymer electrolyte membrane fuel cells, MCFCs do not require an external reformer to convert more energy-dense fuels to hydrogen. Due to the high temperatures at which MCFCs operate, these fuels are converted to hydrogen within the fuel cell itself by a process called internal reforming, which also reduces cost.
Molten carbonate fuel cells are not prone to carbon monoxide or carbon dioxide "poisoning" —they can even use carbon oxides as fuel—making them more attractive for fueling with gases made from coal. Because they are more resistant to impurities than other fuel cell types, scientists believe that they could even be capable of internal reforming of coal, assuming they can be made resistant to impurities such as sulfur and particulates that result from converting coal, a dirtier fossil fuel source than many others, into hydrogen.
The primary disadvantage of current MCFC technology is durability. The high temperatures at which these cells operate and the corrosive electrolyte used accelerate component breakdown and corrosion, decreasing cell life. Scientists are currently exploring corrosion-resistant materials for components as well as fuel cell designs that increase cell life without decreasing performance.
Solid Oxide Fuel Cells
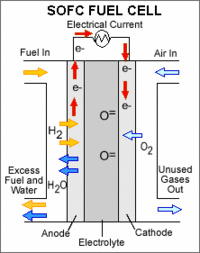 |
An AFC consists of a non-porous metal oxide electrolyte (typically zirconium oxide) sandwiched between an anode (negatively charged electrode) and a cathode (positively charged electrode). The processes that take place in the fuel cell are as follows: 1. Hydrogen fuel is channeled through field flow plates to the anode on one side of the fuel cell, while oxygen from the air is channeled to the cathode on the other side of the cell. 2. At the cathode, a catalyst causes electrons from the electrical circuit to combine with oxygen to create negatively charged oxygen ions. 3. The negatively charged oxygen ions flow through the electrolyte to the anode. 4. At the anode, the catalyst causes the hydrogen to react with the oxygen ions forming water and free electrons. 5. The negatively charged electrons cannot flow through the electrolyte to reach the positively charged cathode, so they must flow through an external circuit, forming an electrical current. 6. At the cathode, the electrons combine with oxygen to create negatively charged oxygen ions, and the process repeats."
Solid oxide fuel cells (SOFCs) use a hard, non-porous ceramic compound as the electrolyte. Because the electrolyte is a solid, the cells do not have to be constructed in the plate-like configuration typical of other fuel cell types. SOFCs are expected to be around 50%–60% efficient at converting fuel to electricity. In applications designed to capture and utilize the system's waste heat (co-generation), overall fuel use efficiencies could top 80%–85%.
Solid oxide fuel cells operate at very high temperatures—around 1,000°C (1,830°F). High-temperature operation removes the need for precious-metal catalyst, thereby reducing cost. It also allows SOFCs to reform fuels internally, which enables the use of a variety of fuels and reduces the cost associated with adding a reformer to the system.
SOFCs are also the most sulfur-resistant fuel cell type; they can tolerate several orders of magnitude more of sulfur than other cell types. In addition, they are not poisoned by carbon monoxide (CO), which can even be used as fuel. This property allows SOFCs to use gases made from coal.
High-temperature operation has disadvantages. It results in a slow startup and requires significant thermal shielding to retain heat and protect personnel, which may be acceptable for utility applications but not for transportation and small portable applications. The high operating temperatures also place stringent durability requirements on materials. The development of low-cost materials with high durability at cell operating temperatures is the key technical challenge facing this technology.
Scientists are currently exploring the potential for developing lower-temperature SOFCs operating at or below 800°C that have fewer durability problems and cost less. Lower-temperature SOFCs produce less electrical power, however, and stack materials that will function in this lower temperature range have not been identified.
Regenerative Fuel Cells
Regenerative fuel cells produce electricity from hydrogen and oxygen and generate heat and water as byproducts, just like other fuel cells. However, regenerative fuel cell systems can also use electricity from solar power or some other source to divide the excess water into oxygen and hydrogen fuel—this process is called "electrolysis." This is a comparatively young fuel cell technology being developed by NASA and others.
Comparison of Fuel Cell Technologies
Each fuel cell technology has advantages and disadvantages. See how fuel cell technologies compare with one another:
- 1000's of Fuel Cell Articles, Video and Audio from The Auto Channel Hydrogen Fuel Cell News Archive
- How Polymer Electrolyte Membrane (PEM) Fuel Cells Work
- How Direct Methanol Fuel Cells Work
- How Alkaline Fuel Cells Work
- How Phosphoric Acid Fuel Cells Work
- How Molten Carbonate Fuel Cells Work
- How Solid Oxide Fuel Cells Work
- How Regenerative Fuel Cells Work
- Comparison of Fuel Cell Technologies
- Comparison Chart (PDF 213 KB) Download Adobe Reader.
Click PLAY to watch the TACH's RoadTrip show about Volkswagen's fuel cell vehicle



